|
1. International Investigator Meetings
Over the past few months, investigators
were brought together on a regional or country level to receive updates and
discuss study progress. We thank all who participated and present some
highlights here.
DSMB Review
The last DSMB Review
took place in early June 2023 and provided valuable data on the participant
cohort. The most common reasons for patient exclusion from study were: indication
for, or contraindication to anticoagulant therapy (306) and patient/family not interested
(261). Investigator feedback indicated that a major source of patient
reluctance for participation was bleeding risk. Strategies for patient
reassurance were discussed:
·
Communicate
low rates of bleeding within study and make patients aware of 3-monthly
DSMB risk monitoring.
·
Build a
relationship of trust between patients and study staff.
·
Explain trial
design is optimised to provide least possible risk to patients through use
of exclusion criteria and monitoring.
Event Rate Improvement
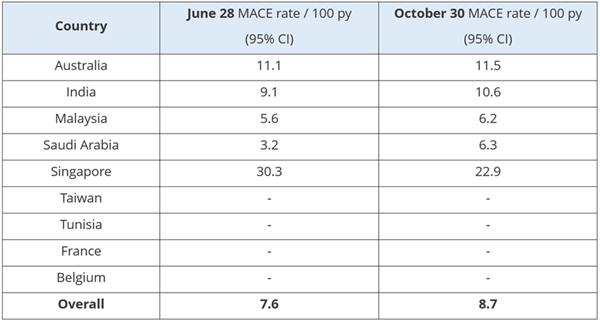
The June DSMB reported a MACE primary
outcome event rate of 6.6
per 100 person years (py), which was
significantly lower than the expected 10 per 100 py
estimated based on previous literature. In the ensuing months, efforts to
improve event-reporting and diligent patient follow-up has yielded a
significant increase to a MACE of 8.7
per 100 py. The MACE rate is expected to rise
further with increased duration of follow-up.
A special
congratulations is extended to Saudi Arabia, who have raised their MACE
rate from 3.2
per 100 py in late June to 6.3 per 100 py through their increased efforts with patient
follow-up. Commendations are also due to India, who have increased their
event rate reporting at some of their highest recruiting sites.
2. Data
Management
Please be reminded that visits should be
conducted within the visit window. Queries are ideally to be to be resolved
within 5-10 days of their creation. We encourage sites to check and resolve
existing queries, with the priority to be given to older entries.
Countries with aged
queries include Malaysia, Australia, Saudi Arabia, Tunisia
and Taiwan. It is our goal to reduce the number of queries as much as
possible by 9th
of November to aid data processing for the
upcoming DSMB
meeting in early December. Resolving queries and completing
missed patient visits will allow sites and staff to start fresh in 2024.
General
Reminders:
·
We have reached 50% of our recruitment goal!
·
Resolve queries and overdue visits if possible by 9th of
November.
·
Tracking of outcomes is essential, check for events
that occur outside of study site or in LTFU participants.
·
Outcome Definitions can be found in Appendix 1 of the
TRACK Protocol: TRACK Trial Endpoint Definitions (page 59)
|